Gas Vesicle (GV) Technology
A Next-Generation Delivery & Vaccine Platform
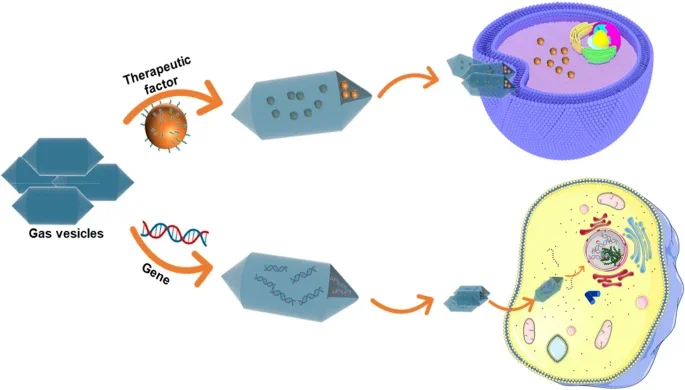
PharmaPlanter leverages its proprietary Gas Vesicle (GV) Platform, an innovative bio-nanotechnology designed for precision drug delivery and highly immunogenic vaccine development. Derived from biological origins, these hollow, proteinaceous nanostructures offer a unique combination of structural stability, biocompatibility, and intrinsic immune-modulating properties.
Our platform enables:
Intrinsic Self-Adjuvanting Feature: Our GVs are engineered to be naturally recognized by the innate immune system. They possess a potent self-adjuvanting effect that triggers robust Antigen-Presenting Cell (APC) activation and T-cell responses without the need for supplemental chemical adjuvants, significantly enhancing the safety and efficacy of vaccine candidates.
Proprietary Multi-Valent Display: Utilizing our advanced genetic engineering platform, we can modularly display a wide array of antigens or functional ligands on the GV surface. This allows for high-density, multi-valent presentation of complex proteins (such as viral spikes or tumor neoantigens), dramatically increasing B-cell receptor cross-linking and antibody titers.
Precision Drug Delivery System: Beyond vaccines, our proprietary GV technology serves as a versatile targeted delivery vehicle. Their unique hollow interior and surface-modifiable shell allow for the encapsulation of small molecules or nucleic acids, enabling protected, site-specific transport and controlled release in challenging physiological environments.
Enhanced Bioavailability & Stability: Unlike lipid-based carriers, GVs are exceptionally resistant to pH changes and enzymatic degradation. This structural resilience ensures that the therapeutic cargo or displayed antigens remain intact and bioactive during systemic circulation, maximizing bioavailability at the target site.
Scalable Biomanufacturing: Our GV platform is optimized for high-yield microbial production, ensuring a cost-effective and highly reproducible manufacturing process. This scalability, combined with our rigorous quality control, facilitates the rapid transition from laboratory-scale innovation to translational research and pilot production.

